Data Integrity at Drug Safety/Pharmacovigilance
Data health has become a critical issue in this era of big data when more pieces of information are processed and stored than ever before. Developing methods that preserve the integrity of the data collected is becoming increasingly important. The first step in keeping data safe is to understand the foundations of data integrity and how it works.
In case the regulatory authority (RA) knocks on the door to inspect the PV/facility onsite, the sudden panic and work disruption would be obvious for some organization. The missing, not in place and not updated critical quality control data would arise many questions during such instances.
Such data accuracy and consistency issues affect all industries and can result in everything from minor inconveniences to major corporate concerns.
Continue reading to find out what data integrity is, why it's important, and what you can do to maintain your data is in good shape.
What is Data Integrity (DI)?
Data integrity is the maintenance and assurance of, data accuracy and consistency over its entire life-cycle and it is a critical aspect to the design, implementation, and usage of any system that stores, processes, or retrieves data. Data integrity followed at each stage of the life cycle of the document (throughout the lifecycle)..
Principles (ALCOA & ALCOA +):
All paper & electronic data to follow the ALCOA+ principles.
ALCOA Attributable, Legible, Contemporaneous, Original and Accurate And the Element “+” Complete, Available, Consistent, Enduring Further details can be referred to the guidance document (GXP’ Data Integrity Guidance and Definitions (March 2018)).
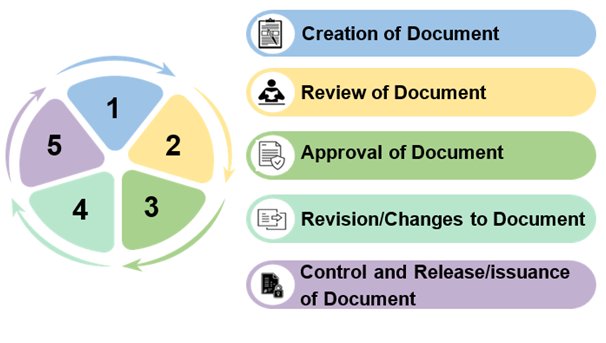
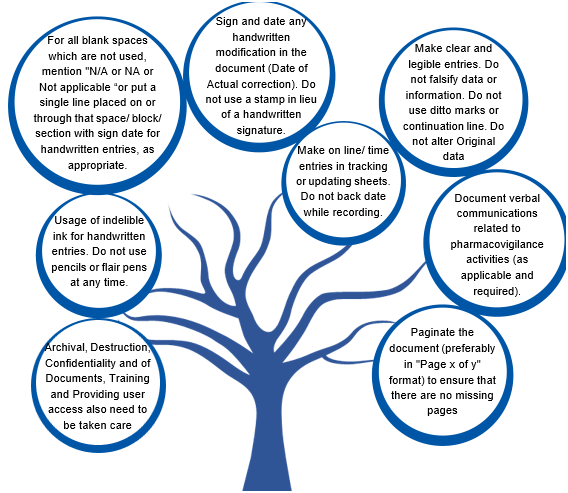
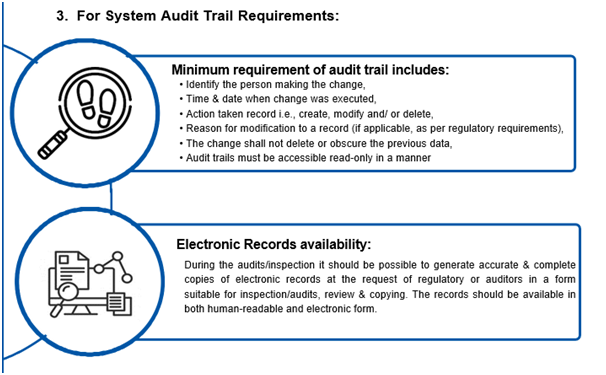
What are the DI rules for the documentation?

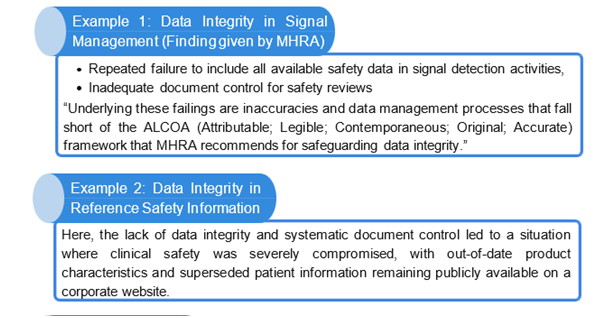
Possible consequences if Data Integrity is not demonstrated at Pharmacovigilance
- Compensation Claims
- MAH authorization cancellation
- Service Providers (CROs) might be banned by Health Authorities
- Reputation damage and Loss of Business
- The site/CRO/Company is not allowed to perform the medical safety activities any more, lead to loss of business
- Legal Prosecution
Let’s look at the possible cause/error which may lead to DI issues in PV (not limited to):
- Too much workload- not included the relevant data
- Deletion of cases from safety data base without proper justification (if the case is falling under late reporting) delete the case
- Agreements related misinformation/ miscommunication
- Mentioning wrong dates intentionally
- Destruction of Source/Original data (forms, reports etc.)
- Change date/time on computer
- Data migration, data storage and security miscondu


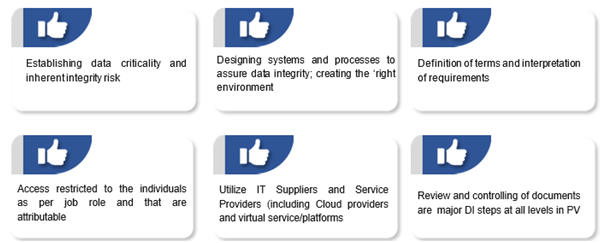
Suggestions to make zero defect DI compliant system