Executive Summary
PROs are defined as “any report of the status of a patient’s health condition that comes directly from the patient, without interpretation by a clinician or anyone else.” These outcomes offer a unique and essential perspective on how patients experience their condition and treatment.PROs encompass:
- Symptoms and side effects(e.g., pain, fatigue, nausea, or anxiety)
- Functional outcomes including physical, emotional, social, sexual, role-based, and cognitive functioning
- Multidimensional constructs such as health-related quality of life (HRQoL) and health utility (Mercieca-Bebber et al., 2018)
Unlike traditional clinical endpoints such as laboratory results or imaging, PROs capture the subjective impact of disease and treatment on a patient’s daily life.
Limitations of Paper-Based PROs
Historically, PRO data were collected using paper diaries and questionnaires. While simple, paper-based methods have significant drawbacks:
- Recall bias: Patients often backfill entries, compromising accuracy.
- Low compliance: Manual diaries lack reminders or time-stamps, with adherence rates as low as 30%.
- Data integrity issues: Handwriting errors, skipped questions, and transcription mistakes reduce reliability.
- Delayed insights and operational burden: Manual collection, entry, and verification slow decision-making and increase cost.
The Transition to ePRO
Electronic PRO (ePRO) systems have transformed data collection by enabling real-time, accurate, and scalable capture of patient-reported information. Key advantages include:
- Improved data quality: Automated validation reduces errors, ensures completeness, and enforces protocol compliance.
- Higher patient engagement: Mobile access, automated reminders, and intuitive interfaces increase adherence.
- Faster access and decision-making: Real-time dashboards allow early detection of adverse events and efficient trial management.
- Regulatory compliance: Systems meet global standards (21 CFR Part 11, GCP) with audit trails, electronic signatures, and standardized data formats.
- Operational efficiency: Reduced manual tasks lower costs, and cloud-based platforms support global scalability.
- Enhanced patient safety and sustainability: Real-time symptom monitoring enables timely clinical intervention, while eliminating paper supports eco-friendly trials.
The Era of Paper Diaries: A Starting Point with Limitations
Early clinical research relied on paper diaries and questionnaires to capture PROs. While simple and accessible, these methods struggled as trials became larger, more complex, and geographically diverse. Paper diaries could not meet the growing need for real-time, high-quality, and scalable data collection, highlighting the need for more efficient, reliable, and patient-friendly solutions.
Key Challenges of Paper-Based PRO Collection
- Recall Bias
Patients frequently completed diary entries retrospectively, leading to significant recall bias. Stone et al. (BMJ 2002) found only 11% of entries were on-time despite 90% self-reported accuracy.This practice “backfilling,” compromises data reliability. - Compliance Monitoring
Lack of automated reminders or time-stamps made adherence hard to track. A notable issue, often referred to as “parking lot syndrome,” involved patients completing multiple diary entries just before a site visit (Bingham et al., 2019). Compliance rates with paper PROs have been reported as low as 30%, compared to 90-97% with ePROs (José and Langel, 2010). - Data Integrity Issues
Handwritten responses were susceptible to transcription errors, illegibility, incomplete entries, and logical inconsistencies. Complex assessments often led to skipped questions or misinterpreted branching logic. Lack of time-stamps permits falsification (Coons et al., 2015). - Delayed Insights
Manual processes such as distributing, collecting, and transcribing paper diaries delays data availability. These inefficiencies hindered timely decision-making and could potentially compromise patient safety or trial outcomes. - Time-Consuming Processes
Paper-based data collection is labor-intensive. Study staff had to prepare, distribute, and manually enter data, while also managing missing or inconsistent entries(Weber et al., 2005). - High Setup and Storage Costs
Maintaining both physical and digital records to meet audit and compliance requirements increase complexity and cost. Scanning, archiving and storage inflate budgets (Meirte et al., 2020; Mercieca-Bebber et al., 2018). - Logistical Risks
Paper diaries completed at home had to be mailed back to study sites, introducing risks such as loss, delays, or non-return of critical data (Mercieca-Bebber et al., 2018).
These limitations drove adoption of electronic solutions that improve integrity, engagement and compliance.
Transition to Digital: The Emergence of ePRO Systems
The adoption of electronic patient-reported outcome (ePRO) systems transformed clinical trial data collection. Initially deployed on handheld devices, ePRO now spans smartphones, tablets, and web platforms, eliminating the limitations of paper while enhancing quality, compliance, and efficiency. As trials became more complex, global, and patient-centric, ePRO emerged as an essential tool for accurate, timely, and scalable data capture, accepted across all trial phases, including post-marketing.

Why ePRO Outperforms Paper
A. Real-Time Data Capture and Reduced Recall Bias
ePRO enables patients to record symptoms at the moment they occur. This eliminates retrospective “back-filling” and significantly reduces recall bias. Automatic time-stamping ensures temporal accuracy, enhancing the reliability of patient-reported data.
B. Enhanced Data Quality and Accuracy
ePRO systems eliminate common errors associated with paper-based methods, such as:
- Illegible handwriting
- Missed fields
- Transcription errors
- Skip pattern inconsistencies
Built-in validation logic, edit checks, and data consistency rules ensure that entries are complete, accurate, and protocol-compliant(Coons et al., 2015).
C. Improved Patient Compliance and Engagement
ePRO boosts adherence via:
- Automated reminders and alerts
- Mobile compatibility for personal devices
- Intuitive survey navigation
These tools reduce patient burden and improve engagement. Studies consistently report higher compliance rates with ePRO (Kiderlen et al., 2023). Push notifications, texts or calls remind patients automatically.
D. Faster Access to Data and Better Decision-Making
Real-time data access allows sponsors and CROs to:
- Identify trends or adverse events promptly
- Intervene clinically when needed
- Adjust trial protocols efficiently
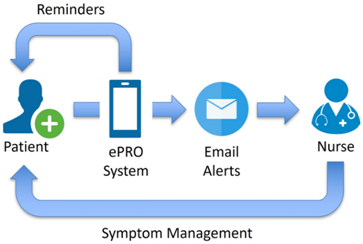
Automation accelerates database lock and interim analyses. Basch et al. (2016)demonstrated that real-time symptom monitoring via ePROs significantly improved overall survival and quality of life for cancer patients. The system triggered email alerts to nurses for severe symptoms, enabling timely clinical interventions.
E. Regulatory Compliance and Data Traceability
ePRO systems are designed to meet global regulatory standards, offering:
- Audit trails
- Automated timestamps
- Electronic signatures
- Standardized data formats
These features ensure traceability and compliance with Good Clinical Practice (GCP) (Zbrozek et al., 2013; Hudgens et al., 2023).
F. Operational Efficiency and Cost Reduction
While ePRO implementation involves upfront investment, it significantly reduces:
- Manual data entry and cleaning
- Source data verification
- Printing, storage, and shipping costs
By automating workflows and minimizing manual tasks, ePRO systems enhance operational efficiency and deliver long-term cost savings.
G. Improved Patient Safety
Real-time symptom reporting enables early detection of safety signals. Investigators can respond promptly to adverse events or deteriorating health, enhancing patient monitoring and clinical intervention. A 2025 meta-analysis of lung-cancer trials found ePRO alerts improved HRQoL (Xia et al.).Clinicians can intervene during critical windows, extending survival.
Source: Mazza, G.L., Dueck, A.C., Ginos, B., Jansen, J., Deal, A.M., Carr, P., Blinder, V.S., Thanarajasingam, G., Jonsson, M., Lee, M.K. and Rogak, L.J., 2024. Optimization of alert notifications in electronic patient-reported outcome (ePRO) remote symptom monitoring systems (AFT-39). Quality of Life Research, 33(7), pp.1985-1995.
H. Completeness and Timeliness of Data Collection
ePRO platforms ensure that all required fields are completed before submission. Features like logic-based question flows, built-in instructions, and automated prompts improve:
- Data completeness
- Timeliness
- Monitoring efficiency
I. Environmentally Sustainable
By replacing paper-based methods, ePRO systems support eco-friendly trial operations. Digital workflows eliminate the need for printing, shipping, and physical storage, reducing waste and carbon footprint aligning with sustainability goals and digital transformation initiatives in life sciences(Pawar et al., 2017).
Comparison between paper-based PROs and Electronic PROs
| Paper-Based PROs (pPROs) | Electronic PROs (ePROs) | |
|---|---|---|
| Data Collection | Handwritten on printed forms | Entered via electronic devices (phone, tablet, web etc.) |
| Data Accuracy & Quality | Prone to human errors (e.g., handwriting, transcription), backfilling, and recall bias | Real-time entry, edit checks and forced fields cut errors |
| Timeliness & Availability | Data access is delayed due to manual handling and entry | Real-time data capture and instant availability for analysis |
| Patient Compliance | Lower compliance; dependent on memory and motivation | Higher compliance with automated reminders and easier access |
| Monitoring & Oversight | Limited or delayed oversight; real-time issues often go unnoticed | Enables real-time remote monitoring and early intervention |
| Patient Experience | Less convenient; patients must carry forms and remember timing | More user-friendly; can use personal devices at any time |
| Accessibility & Inclusion | More accessible for non-digital users; no internet or device needed | May exclude users with low digital literacy or no device/internet access |
| Administrative Effort | High; includes distribution, collection, transcription, and storage | Reduced; automation minimizes manual work and errors |
| Cost | Lower initial cost; higher over time due to labor and storage | Higher set-up cost, lower total cost |
| Multilingual | Requires separate forms per language | Supports multiple languages in one system |
| Data Management & Reporting | Manual entry, storage, and analysis; time-consuming | Automated capture, storage, and real-time analytics and reporting |
| Scalability | Limited scalability; resource-intensive for multi-site studies | Highly scalable across regions, sites, and geographies |
| Implementation Considerations | Familiar workflow; minimal tech needs but resistant to change | Needs training, devices, connectivity |
Regulatory Endorsement and Industry Standards
FDA and EMA guidance now recommend validated ePRO systems. Key considerations include:
- 21 CFR Part 11 Compliance:Ensures requirements for electronic records and electronic signatures. Compliant systems provide audit trails, user authentication and data-integrity controls.” (eCFR, USFDA, 2025).
- FDA PRO Guidance (2009): Provides direction for selecting, developing, and interpreting PRO tools in support of labelling claims. It emphasizes the need for validated instruments and encourages electronic collection methods to enhance data quality and traceability (PRO guidance, USFDA, 2009).
- ISPOR and CDISC Standards:
- ISPOR (International Society for Pharmacoeconomics and Outcomes Research): Provides best practices for PRO instrument development and validation, ensuring equivalence between paper and electronic modes and regulatory compliance (Coons et al., 2009; Zbrozek et al., 2013).
- CDISC (Clinical Data Interchange Standards Consortium): Defines standardized models for ePRO data collection, analysis, and submission, improving traceability, interoperability, and audit-readiness(Hudgens et al., 2023).
The Modern ePRO Landscape
As clinical trials evolve toward decentralized, patient-centric, and technology-enabled models, ePRO systems have matured from simple digital diaries into comprehensive electronic Clinical Outcome Assessment (eCOA) platforms.
Key Innovations Driving Modern ePRO Systems
-
Bring Your Own Device (BYOD)
BYOD lets participants use their own phones or tablets.This approach:- Enhances patient convenience and familiarity
- Improves engagement and compliance
- Reduces hardware provisioning costs for sponsors
-
Wearable and Sensor Integration
ePRO systems now integrate with wearables and remote sensors, combining subjective patient-reported data with objective physiological metrics such as heart rate, activity levels, sleep quality, or oxygen saturation.
This fusion of real-world data (RWD) with PROs enhances the depth and clinical relevance of trial insights. The WEAR study showed this fusion clarifies RA disease activity (Stradford 2024).
-
Multilingual and Cross-Cultural Support
With the globalization of clinical trials, modern ePRO systems helps to ensure:- Linguistic and contextual appropriateness
- Regulatory compliance for global submissions
- Inclusivity across diverse patient populations
-
Cloud-Based Dashboards and Real-Time Analytics
Advanced ePRO platforms utilize cloud technologies to deliver:- Real-time access to patient-reported data
- Interactive dashboards for sponsors and sites
- Risk-based monitoring tools to flag compliance issues, protocol deviations, or safety concerns
Navigating ePRO Implementation: Challenges and Mitigation Strategies
While ePRO systems offer substantial benefits in terms of data quality, patient centricity, and operational efficiency, their implementation presents a range of challenges. These span technical, operational, human, and regulatory domains, each requiring thoughtful planning, risk mitigation, and cross-functional collaboration to ensure successful deployment.
A. Patient Access and Digital Literacy
Challenges:
- Device gap: many patients lack a smartphone or tablet. This may require sponsors to provision devices, adding logistical and financial complexity.
- Digital Literacy: Older adults or patients from underserved communities may lack familiarity with mobile technology. Without proper onboarding, this can lead to non-compliance and compromised data quality.

Mitigation Strategies:
- Provide pre-configured devices with support to ensure access and data quality.
- Design intuitive user interfaces with simple navigation.
- Provide multi lingual training materials (e.g., videos, guides).
- Offer caregiver or site-level support when needed.
B. Technical and Logistical Complexity
Challenges:
- Device Management: Provisioned models require procurement, distribution, maintenance, and replacement of devices, placing strain on site and sponsor resources.
- Connectivity Issues: Inconsistent cellular or Wi-Fi coverage, especially in rural or remote areas, can delay data transmission or interrupt assessments.
- System Support: Technical failures or software bugs must be resolved quickly to prevent data loss or patient dropout.

Mitigation Strategies:
- Use centralized device logistics to streamline handling and reduce site burden.
- Conduct site-level feasibility assessments for connectivity.
- Use offline-capable systems that sync when connectivity is restored
- Establish 24/7 technical support for patients and sites.
C. Data Security, Privacy, and Compliance
ePRO systems must comply with stringent data protection regulations, including, GDPR (EU), HIPAA and 21 CFR Part 11 (US).
Challenges:
- Data breaches can erode patient trust and lead to legal consequences.
- Non-compliance may result in regulatory findings or trial delays.

Mitigation Strategies:
- Implement encryption, user authentication, and audit trails.
- Ensure vendors follow documented validation and compliance protocols.
- Conduct privacy impact assessments during planning.
D. System Validation and Regulatory Readiness
Before deployment, ePRO systems must be validated to demonstrate reliability, accuracy, and audit-readiness, especially for trials intended for regulatory submission.
Challenges:
- Validation requires time, expertise, and alignment with sponsor Quality Management Systems (QMS).
- User Acceptance Testing (UAT) must simulate real-world use scenarios.

Mitigation Strategies:
- Engage experienced QA and IT teams for validation.
- Use standardized validation templates and protocols.
- Select vendors that can supply prior 483s or inspection success letters.
E. Cost and Resource Allocation
Challenges:
- Upfront Investment: Licensing, device procurement (if not BYOD), staff training, translations, and infrastructure.
- Ongoing Costs: Maintenance, upgrades, patient support, and data monitoring must be factored into long-term budgets.

Mitigation Strategies:
- Model 3-year ROI: fewer queries, faster database lock, less SDV
- Explore cost-saving options like BYOD or reusable device inventories.
- Align internal stakeholders on budget early in the study design phase.
F. End-User Engagement and Acceptance
Challenges:
- Staff Buy-In: Investigators and coordinators may resist workflow changes introduced by ePRO systems.
- Patient Fatigue: Frequent or lengthy assessments can lead to disengagement and missing data.

Mitigation Strategies:
- Set clear expectations with sites and stakeholders early.
- Communicate the clinical and operational benefits of ePRO.
- Incorporate visual progress tracking or feedback tools to motivate patients.
Proactive mitigation turns these barriers into competitive advantages and protects the patient experience.
Conclusion
The evolution from paper diaries to ePRO systems represents a critical advancement in patient-centric clinical research. By combining high-quality data capture, regulatory compliance, and patient engagement, ePRO technology strengthens trial integrity, operational efficiency, and clinical decision-making. Organizations adopting validated, user-friendly, and scalable ePRO solutions are better positioned to conduct inclusive, efficient, and impactful trials that truly reflect patient needs.
The transition from paper diaries to ePRO systems represents a significant advancement in clinical research. By enabling real-time data capture, improving compliance, and enhancing data quality, ePRO technology has reshaped how patient experiences are measured and understood. As trials become decentralized and patient-centric, ePRO systems are essential for ensuring data integrity, regulatory compliance, and meaningful patient engagement. Their seamless integration with modern technologies and global scalability makes them indispensable in today’s research landscape.
Organizations that invest in scalable, validated, and user-friendly ePRO solutions are better positioned to conduct high-quality, efficient, and inclusive clinical trials, ultimately accelerating the development of therapies that truly reflect patient needs and outcomes.
References
- Basch, E., Deal, A.M., Kris, M.G., Scher, H.I., Hudis, C.A., Sabbatini, P., Rogak, L., Bennett, A.V., Dueck, A.C., Atkinson, T.M. and Chou, J.F., 2016. Symptom monitoring with patient-reported outcomes during routine cancer treatment: a randomized controlled trial. Journal of Clinical Oncology, 34(6), pp.557-565.
- Basch, E., Schrag, D., Henson, S., Jansen, J., Ginos, B., Stover, A.M., Carr, P., Spears, P.A., Jonsson, M., Deal, A.M. and Bennett, A.V., 2022. Effect of electronic symptom monitoring on patient-reported outcomes among patients with metastatic cancer: a randomized clinical trial. Jama, 327(24), pp.2413-2422.
- Basch, E., Schrag, D., Jansen, J., Henson, S., Ginos, B., Stover, A.M., Carr, P., Spears, P.A., Jonsson, M., Deal, A.M. and Bennett, A.V., 2025. Symptom monitoring with electronic patient-reported outcomes during cancer treatment: final results of the PRO-TECT cluster-randomized trial. Nature medicine, pp.1-8.
- Billingy, N.E., Tromp, V.N., Aaronson, N.K., Hoek, R.J., Bogaard, H.J., Onwuteaka-Philipsen, B.D., van de Poll-Franse, L., Hugtenburg, J.G., Belderbos, J., Becker-Commissaris, A. and van den Hurk, C.J., 2023. Quality of life after patient-initiated vs physician-initiated response to symptom monitoring: the SYMPRO-Lung trial. JNCI: Journal of the National Cancer Institute, 115(12), pp.1515-1525.
- Billingy, N.E., van den Hurk, C.J., Tromp, V.N., van de Poll-Franse, L., Onwuteaka-Philipsen, B.D., Hugtenburg, J.G., Bogaard, H.J., Belderbos, J., Aaronson, N.K., Walraven, I. and Becker-Commissaris, A., 2024. Patient-vs physician-initiated response to symptom monitoring and health-related quality of life: the SYMPRO-lung cluster randomized trial. JAMA Network Open, 7(8), pp.e2428975-e2428975.
- Bingham III, C.O., Gaich, C.L., DeLozier, A.M., Engstrom, K.D., Naegeli, A.N., De Bono, S., Banerjee, P. and Taylor, P.C., 2019. Use of daily electronic patient-reported outcome (PRO) diaries in randomized controlled trials for rheumatoid arthritis: rationale and implementation. Trials, 20(1), p.182.
- Coons, S.J., Eremenco, S., Lundy, J.J., O’Donohoe, P., O’Gorman, H. and Malizia, W., 2015. Capturing patient-reported outcome (PRO) data electronically: the past, present, and promise of ePRO measurement in clinical trials. The Patient-Patient-Centered Outcomes Research, 8(4), pp.301-309.
- Coons, S.J., Gwaltney, C.J., Hays, R.D., Lundy, J.J., Sloan, J.A., Revicki, D.A., Lenderking, W.R., Cella, D. and Basch, E., 2009. Recommendations on evidence needed to support measurement equivalence between electronic and paper‐based patient‐reported outcome (PRO) measures: ISPOR ePRO Good Research Practices Task Force report. Value in Health, 12(4), pp.419-429.
- Dickson, N.R., Beauchamp, K.D., Perry, T.S., Roush, A., Goldschmidt, D., Edwards, M.L. and Blakely, L.J., 2022. Real-world use and clinical impact of electronic patient-reported outcomes (ePROs) in patients with solid tumors treated with immuno-oncology (IO) therapy.
- Food and Drug Administration (US). Patient-reported outcome measures: use in medical product development to support labeling claims. 2009 Dec 9. Available from: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/patient-reported-outcome-measures-use-medical-product-development-support-labeling-claims
- Generalova, O., Roy, M., Hall, E., Shah, S.A., Cunanan, K., Fardeen, T., Velazquez, B., Chu, G., Bruzzone, B., Cabot, A. and Fisher, G.A., 2021. Implementation of a cloud-based electronic patient-reported outcome (ePRO) platform in patients with advanced cancer. Journal of Patient-Reported Outcomes, 5(1), p.91.
- Hudgens, S., Kern, S., Barsdorf, A.I., Cassells, S., Rowe, A., King-Kallimanis, B.L., Coon, C., Low, G. and Eremenco, S., 2023. Best practice recommendations for electronic patient-reported outcome dataset structure and standardization to support drug development. Value in Health, 26(8), pp.1242-1248.
- José, N.C. and Langel, K., 2010. ePRO vs. Paper. Applied Clinical Trials, 19(6).
- Kiderlen, T.R., Schnack, A. and de Wit, M., 2023. Essential barriers and considerations for the implementation of electronic patient-reported outcome (ePRO) measures in oncological practice: contextualizing the results of a feasibility study with existing literature. Journal of Public Health, 31(12), pp.2071-2088.
- Meirte, J., Hellemans, N., Anthonissen, M., Denteneer, L., Maertens, K., Moortgat, P. and Van Daele, U., 2020. Benefits and disadvantages of electronic patient-reported outcome measures: systematic review. JMIR perioperative medicine, 3(1), p.e15588.
- Mercieca-Bebber, R., King, M.T., Calvert, M.J., Stockler, M.R. and Friedlander, M., 2018. The importance of patient-reported outcomes in clinical trials and strategies for future optimization. Patient related outcome measures, pp.353-367.
- Pawar, S.G., Ramani, P.S., Prasad, A., Dhar, A., Babhulkar, S.S. and Bahurupi, Y.A., 2017. Software version of Roland Morris Disability questionnaire for outcome assessment in low back pain. Neurological research, 39(4), pp.292-297.
- Philipps, L., Foster, S., Gardiner, D., Gillman, A., Haviland, J., Hill, E., Manning, G., Stiles, M., Hall, E. and Lewis, R., 2022. Considerations when introducing electronic patient-reported outcome data capture in multicentre oncology randomised controlled trials. Trials, 23(1), p.1004.
- Rivera, S.C., Kyte, D.G., Aiyegbusi, O.L., Slade, A.L., McMullan, C. and Calvert, M.J., 2019. The impact of patient-reported outcome (PRO) data from clinical trials: a systematic review and critical analysis. Health and quality of life outcomes, 17(1), p.156.
- Soto-Rey, I., Rehr, M., Bruland, P., Zeidler, C., Riepe, C., Steinke, S., Ständer, S., Dugas, M. and Storck, M., 2018. Electronic collection of multilingual patient-reported outcomes across Europe. Methods of Information in Medicine, 57(S 02), pp.e107-e114.
- Stone, A.A., Shiffman, S., Schwartz, J.E., Broderick, J.E. and Hufford, M.R., 2002. Patient non-compliance with paper diaries. Bmj, 324(7347), pp.1193-1194.
- Storck, M., Christians, G., Möller, B., Dugas, M. and Soto-Rey, I., 2018. Conducting a multilingual study researching traumatised refugees utilizing a patient-reported outcome system. In German Medical Data Sciences: A Learning Healthcare System (pp. 109-113). IOS Press.
- Stradford, L., Curtis, J.R., Zueger, P., Xie, F., Curtis, D., Gavigan, K., Clinton, C., Venkatachalam, S., Rivera, E. and Nowell, W.B., 2024. Wearable activity tracker study exploring rheumatoid arthritis patients’ disease activity using patient-reported outcome measures, clinical measures, and biometric sensor data (the wear study). Contemporary Clinical Trials Communications, 38, p.101272.
- Thukral, A., Linsmeier, K., Fowler, B. and Bhardwaj, S., 2022. Bring your own device: a practical framework to leverage in the electronic patient-reported outcomes data collection in clinical trials. Journal of the Society for Clinical Data Management, 1(4).
- U.S. Food and Drug Administration. 21 CFR Part 11-Electronic Records; Electronic Signatures. Available from: https://www.ecfr.gov/current/title-21/chapter-I/subchapter-A/part-11.
- Weber, B.A., Yarandi, H., Rowe, M.A. and Weber, J.P., 2005. A comparison study: paper-based versus web-based data collection and management. Applied Nursing Research, 18(3), pp.182-185.
- Xia, Y., Guan, X., Zhu, W., Wang, Y., Shi, Z. and He, P., 2025. Effectiveness of symptom monitoring on electronic patient-reported outcomes (ePROs) among patients with lung cancer: a systematic review and meta-analysis. npj Digital Medicine, 8(1), p.399.
- Zbrozek, A., Hebert, J., Gogates, G., Thorell, R., Dell, C., Molsen, E., Craig, G., Grice, K., Kern, S. and Hines, S., 2013. Validation of electronic systems to collect patient-reported outcome (PRO) data—recommendations for clinical trial teams: report of the ISPOR ePRO Systems Validation Good Research Practices Task Force. Value in Health, 16(4), pp.480-489.
